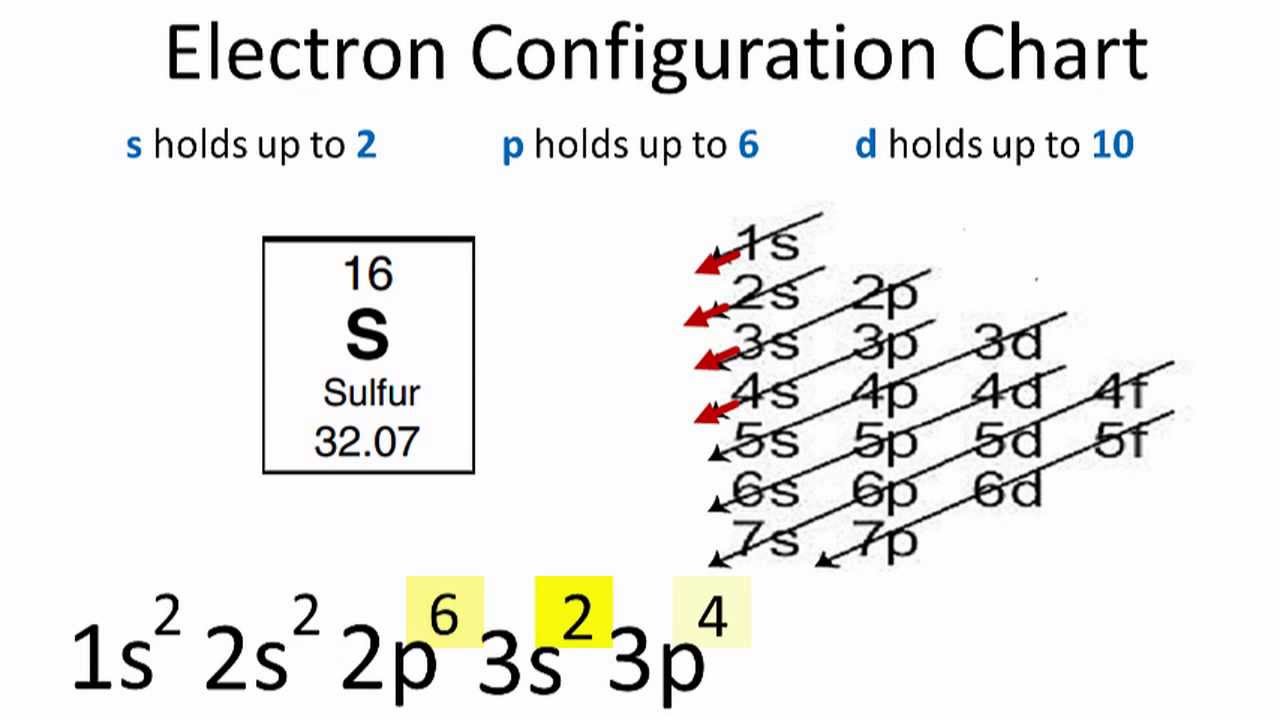
The remaining 4 electrons must be shown as normal. This takes care of the first 10 electrons (the atomic number of neon). The noble gas must have few electrons than the element we are working with. But, we must go back to find the closest noble gas. Ne 3s2 3p2 The full electron configuration of silicon is 1s2 2s2 2p6 3s2 3p2.To find its noble gas shorthand, look at the full electron configuration and see how many filled shells there are.For silicon, all but the outermost shell is filled. Notice that argon is closer to silicon than neon. To write the noble gas configuration, find the noble gas that is just before silicon on the periodic table. A schematic diagram of the designed all-silicon metasurface based on CP multiplexing encoding technique is shown in Figure 1, which can produce the off-axis tightly focused beams carrying specific polarization information in the focal plane 2830. The number of electrons in K orbit 2 (1) 2 2. Using the diagonal rule, the electron configuration for silicon looks like this: 1s 22s 22p 63s 23p 2 The condensed electron configuration, also known as the noble gas configuration, for lithium is: 2s 1 After the noble gas, continue on with the valence electrons. In the case of lithium, represents the 1s 2 electrons. Each added electron is assigned to the lowest-energy sublevel available. This noble gas in brackets represents all of the electrons that are not in the outer shell, the core electrons. To determine the electron configuration of a particular atom, start at the nucleus and add electrons one by one until the number of electrons equals the number of protons in the nucleus. In this case, helium is the noble gas that is just before lithium. So, 3 electrons of Gallium atom will form covalent bonds with 3 electrons of Silicon atom. To write the condensed configuration, find the noble gas that occurs on the periodic table just before the atom. The electron configurations of silicon (14 electrons), phosphorus (15 electrons), sulfur (16 electrons), chlorine (17 electrons), and argon (18 electrons) are analogous in the electron configurations of their outer shells to their corresponding family members carbon, nitrogen, oxygen, fluorine, and neon, respectively, except that the principal. A condensed electron configuration shows the outermost electrons. The outermost electrons, also known as the valence electrons, are the ones that are the most important to chemists, since they are the electrons involved in chemical reactions. Some electron configurations can get quite long. Electron configurations can get very long, as you can see in the table above A shortened form of the electron configuration can be written for most. When writing electron configurations, sometimes it is beneficial to have a shorthand method.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
